The mineral group called zeolites is incredibly useful. It got its name back in the 1700s, when the Swedish mineralogist who also discovered nickel saw steam billowing out of a rock he was heating. Zeo means “boiling” and lithos means “stone.”
That water had gotten into that rock – which was the mineral stilbite – in the first place thanks to the porous structure and electrical charge that all zeolite group members have.
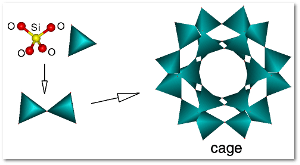
What is a zeolite?
Every zeolite molecule has a metal atom (usually silicon or aluminum) sitting in the middle of a tetrahedon with four oxygen atoms at each apex.
These tetrahedrons stack up to form channels that come in very handy when a tiny filter is needed, say, to sieve out impurities in petroleum or natural gas or to strain toxins out of water or air.
The water that Swedish scientist boiled out of his stilbite specimen was stored in these channels.
In man-made zeolites, the diameter of the channels can be adjusted to suit whatever purpose the zeolite is going to serve. For example, in oxygen generation, it can be made just large enough to filter oxygen out of the air.
Electrical charge is important, too.
When silicon is at the center of a zeolite tetrahedon there is no charge, but if it’s aluminum, the overall charge is negative.
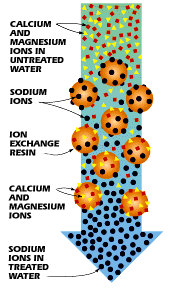
That draws in positively charged metal ions (sodium, magnesium, chloride, calcium, and others) or water.
This is great when you need to dry something out or change its chemical composition (like softening “hard” water).
How do zeolites form?
In nature, you’ll find zeolites in areas where molten rock or hot volcanic ash and alkaline water have met. Of course, there’s an explosion when that happens. This leaves air pockets inside the igneous rock where zeolites and other minerals can form.
A slower way to make natural zeolites is to just let shallow water sit. These minerals sometimes will crystallize out, but it takes thousands or millions of years.
However they form, zeolites are collected via open-pit mining, especially in China, South Korea, Japan, Jordan, Turkey, and the US.
Natural zeolite comes mixed in with a lot of other materials that have to be sorted out. This sales video shows a mining operation (it’s a random video found on YouTube – I’m not connected with the company in any way and know nothing about it).
That’s a lot of work and energy, so they have figured out how to make pure zeolites in the lab.
Take aqueous solutions of aluminum oxide and silicone oxide. Add sodium hydroxide (or sodium aluminate or sodium silicate) and…stir well?

No, the actual process, called sol-gel, is more complex. It produces different results based on the composition of the mixture (ammonia or other positively charged ions can be added), as well as its pH, temperature, and timing.
By controlling those factors, we can even design zeolites unlike any found in nature.
There are about 40 known natural zeolite structures, and over 200 artificial ones, with more being added all the time. The International Zeolite Association database lists approved structures.
Can zeolites cure my …?
There are some amazing alternative-medicine claims for zeolite. Supposedly they’re helpful in treating cancer, autism, diarrhea and herpes. Zeolites also allegedly balance body pH and rid the body of heavy metals.

Although zeolite is used in livestock feed to absorb toxins, there is no objective proof to back up any of those claims for humans. The FDA has been going after some online marketers.
Zeolites may even be harmful. Elevated levels of arsenic, lead, mercury, cadmium, nickel, copper, and chromium have been found in a European zeolite cure-all. There is evidence that zeolites may interfere with medications when taken orally. They can even cause a very horrid condition called graft-versus-host disease in immune-suppressed patients.
Worst of all, zeolite dust, if inhaled, has been linked to malignant mesothelioma, according to the Memorial Sloan Kettering Cancer Center.
It may be hazardous even to live near an outcrop of one of the members of the zeolite group called erionite.
That said, hospitals do use zeolites in a variety of ways, including as a dressing treatment to stop heavy bleeding – they draw out the water component of blood, leaving behind the blood’s clotting materials. Other medical uses include acting as a suspension in some medications and during cardiopulmonary bypass and hemodialysis procedures.
What else are zeolites used for?
Thomsonite, the only gemstone variety of zeolite, is rare, but it has been found on every continent but Australia.

Porous structure and the ease with which they exchange ions make zeolites useful in many different industrial applications.
The ones packed into your powdered laundry detergent soften water by replacing calcium ions in hard water with sodium ions. Phosphates used to do this, but the switch was made when people realized phosphates harm the envirionment.
Zeolites are also good catalysts. Their pore size and acidity can be adjusted as needed.
For example, did you ever wonder how that gooey petroleum in the ground gets turned into something useful? Zeolites are major players in the production of gasoline, LPG gas, jet fuel, diesel, and naphtha through catalytic cracking.
Basically, a long hydrocarbon molecule is broken down into many smaller and simpler molecules in the presence of a catalyst.
Here’s a video, apparently made by a silent movie aficionado, that shows how catalysts (crushed pumice stone and clay pots in the video, zeolites in industry) turn hydrocarbons into flammable gas and other products.
(Of note, alkenes help other petrochemical industrial processes along.)
Sigh. I would have done much better in high school chemistry lab if it had had a soundtrack like that.
The above two things – water softening in detergents and catalytic cracking in the oil industry – are the top two uses of zeolites worldwide.
There are other ways to take advantage of their porous structure. Zeolites filter out odors, toxins and chemicals. Most municipal water suppliers use zeolite filters.
A synthetic zeolite’s pore size can even be designed to filter nuclear waste, trap it in the pore spaces, and then store that waste as stone. This is much more stable tomb for radioactivity than a barrel.
According to Amethyst Galleries, zeolites can also be used for molecular transport (think extended water delivery to plant roots, for instance, or medication to a specific part of the body):
Zeolites can have the water in their structures driven off by heat with the basic structure left intact. Then other solutions can be pushed through the structure…This process has applications in medicine, livestock feeds and other types of research.
It’s nice that this incredibly valuable material can by easily made out of silicon and aluminum – two of the most abundant elements in Earth’s crust. Zeolites are useful for many different things, from cleaning laundry to storing nuclear waste.
Sources:
- The Zeolite Group of Minerals. Amethyst Galleries
- Zeolites. IHS Chemical
- Zeolites. United States Geologic Survey (PDF)
- Zeolite. Wikipedia
